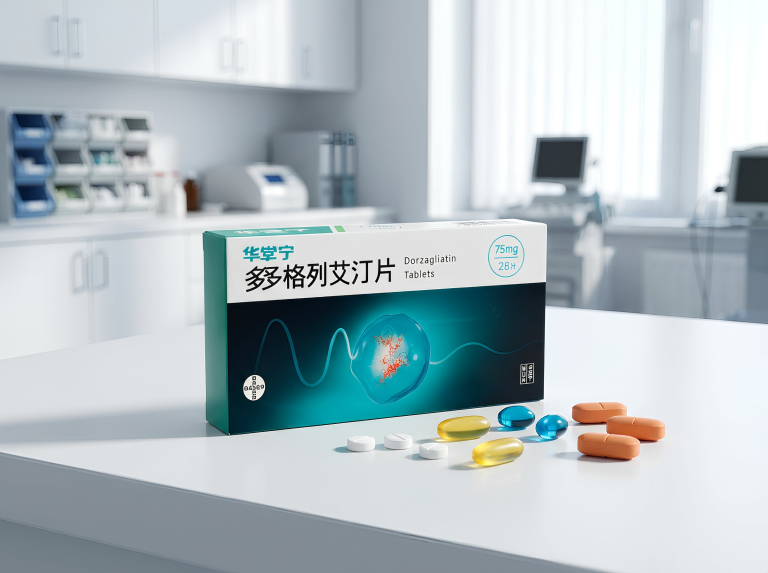
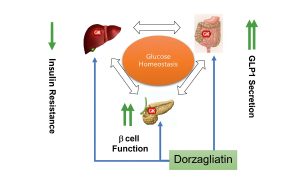
Current treatment of type 2 diabetes requires not only rapid reduction of glucose levels, but also long-term stability, restoration of beta-cell function and minimization of risks. In this context, Dorzagliatin — a first — in-class dual glucokinase activator, has shown impressive results in both short-and long-term therapy. Data from 24-and 52-week studies allow us to judge its potential as a drug that affects not only the symptoms, but also the mechanism of the disease itself.
Short-term effectiveness: data from the 24-week SEED study
The SEED clinical trial, performed according to the principles of randomization and double-blind control, evaluated the effectiveness of Dorzagliatin monotherapy for 24 weeks. The study included patients with insufficient glycemic control during baseline treatment.
The results were convincing: the average reduction in the level of glycated hemoglobin (HbA1c) was 1.15% compared to 0.58% in the placebo group. This result reflects not just a temporary effect, but the rapid inclusion of the drug in the pathogenetic chain of sugar regulation. After 4 weeks of therapy, the majority of participants showed positive dynamics of glycemic indicators.
The effect of Dorzagliatin on postprandial glucose and beta-cell function deserves special attention. By activating glucokinase, the drug enhances the insulin response in the first hours after eating, reducing glycemic peaks. At the same time, the functional activity of β-cells improves, which is confirmed by an increase in HOMA2-β parameters. This indicates not only a symptomatic, but also a pathogenetic effect that contributes to a decrease in the metabolic load on the pancreas.
The safety profile was favorable: the incidence of hypoglycemia was less than 1%, and no serious adverse events were reported.
Proof of stability: 52-week DREAM study
If short courses of therapy show effectiveness, then long-term follow-ups are important for assessing the stability of the effect and the potential for remission. The DREAM study, which lasted 52 weeks, provided answers to these questions.
This observational study included patients with T2DM who received Dorzagliatin for one year without additional hypoglycemic treatment. At the end of the course, 65.2% of patients kept the HbA1c level below 7%, which corresponds to the target range of most international recommendations. This achievement is particularly important given the lack of auxiliary drugs: Dorzagliatin actually maintained remission of the disease in monotherapy.
Such data can be considered as signs of deep metabolic correction: Dorzagliatin not only compensates for sugar, but also stabilizes the function of the pancreas, reduces insulin resistance, and helps restore homeostasis without increasing the risk of hypoglycemia. This reflects a qualitatively new approach in the treatment of diabetes-the transition from controlling symptoms to controlling the progression of the disease.
Clinical significance and application prospects
The results of the SEED and DREAM studies demonstrate that Dorzagliatin combines three key qualities of the ideal hypoglycemic drug:
- Quick action: clinically significant reduction in HbA1c after just 4 weeks.
- Sustainability: maintaining glycaemia in the target range for 52 weeks without additional medications.
- Security: minimal risk of hypoglycemia, good tolerability, no severe side effects.
These characteristics make Dorzagliatin a relevant choice for patients with inadequate metformin compensation, as well as for monotherapy in the early stages of T2DM. In addition, it opens up the prospect of a strategy of metabolic remission — when the disease is brought under control not by constant pharmacological pressure, but by restoring the regulatory functions of the body.
Dorzagliatin demonstrated significant and reproducible results in clinical trials lasting 24 and 52 weeks. It quickly reduces glucose levels, stabilizes patients ‘ condition, and maintains its effectiveness in the long run. Its safety profile and its effect on beta-cell function make it one of the most promising next-generation drugs for the treatment of type 2 diabetes.
Sources:
https://www.huamedicine.com
https://pmc.ncbi.nlm.nih.gov
https://diabetesjournals.org/diabetes
https://www1.hkexnews.hk
https://onlinelibrary.wiley.com