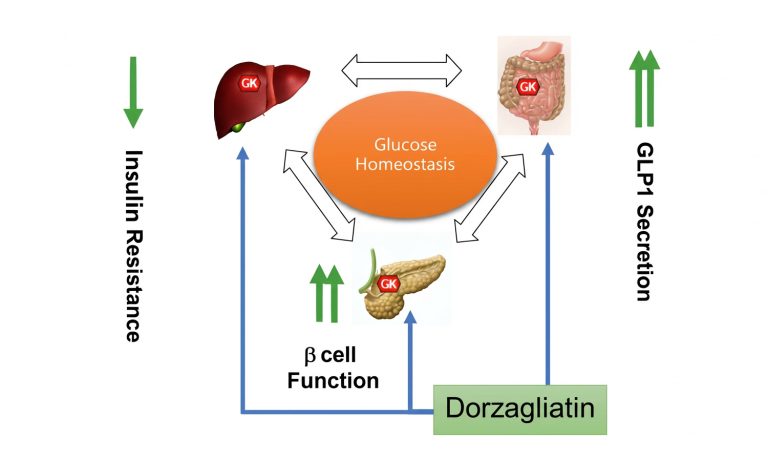
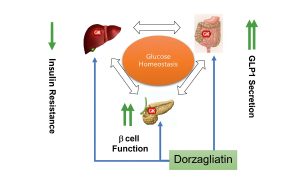
In the modern therapy of type 2 diabetes mellitus (T2DM), drugs that not only reduce glucose levels, but do it quickly, stably and with minimal risk of complications are becoming increasingly important. .Dorzagliatin, a new generation of glucokinase activator, has demonstrated just such qualities in a number of large clinical studies. It activates the metabolic pathways responsible for the production of insulin and the control of glucose production by the liver, restoring the functioning of key organs: the pancreas and liver.
Clinical trials: DAWN, SEED and HMM0302
DAWN study (Phase III, combination with metformin)
Objective: to evaluate the efficacy and safety of Dorzagliatin in combination with metformin in patients with insufficient DM2 control.
24-week results:
- HBA₁C reduction: -1.02 % in the Dorsagliatine + metformin group vs. -0.36 % in the placebo group;
- Achievement of the HBA₁C target < 7.0 % was achieved in 44.4 % of patients versus 10.7 % in the control group;
- Improvement of β-cell function (by HOMA2-β);
- Hypoglycemia was observed in less than 1 % of cases;
- Effect Speed: the sugar reduction started as early as the 4th week and continued throughout the entire period.
SEED Study (monotherapy)
The effectiveness of Dorzagliatin without additional hypoglycemic agents was evaluated.
24-week results:
- HBA₁C reduction: -1.15 % vs. -0.58 % in the placebo group;
- High self-efficacy is confirmed;
- Good security and portability profile.
Study HMM0302 (supplement to metformin)
- Stable reduction of HBA₁C and improvement of postprandial glycemia;
- Early and stable dynamics against the background of already ongoing metformin therapy;
- The drug enhances the effect without the need to increase the dosage of the base drug.
Meta-analysis and aggregated data
A meta-analysis (2022) based on data from 1,332 patients from randomized clinical trials showed:
- HBA₁C reduction: average −0.65 % compared to placebo (95 % CI: -0.76 … -0.54);
- Reduced Fasting Glucose (FPG): an average of 9.2 mg / dl is especially important for controlling morning glycemia;
- Postprandial glucose (2h-PPG): decreased by -48.7 mg / dl, which means better compensation after meals;
- Growth of beta-cell function: the HOMA2-β index increased by +2.69, which confirms an improvement in insulin secretion;
- Security Profile: without increasing the risk of serious side effects; hypoglycemia is extremely rare.
These data confirm that Dorzagliatin has not only statistical, but also clinically significant efficacy in various therapeutic regimens.
What does this mean for patients
What is important for a diabetic patient is not just a reduction in sugar, but a tangible improvement in well-being, predictability of treatment, and reduced risks.
Here’s how it feels in real life:
- Fast effect. After 3-4 weeks, morning and afternoon sugars begin to decrease. This reduces fatigue, irritability, and dry mouth.
- Feel better after eating. There is no sudden drowsiness, headache, or pressure spikes — Dorzagliatin controls postprandial peaks.
- Clear morning. The fasting glucose level stabilizes — leaving you feeling tired and having headaches when you wake up.
- Reducing anxiety. The minimal risk of hypoglycemia relieves the fear of sudden sugar drops-especially at night.
- Physiological help. Dorzagliatin does not just’ crush ‘ glucose, but helps the pancreas to work properly-as at the beginning of the disease.
- Fewer visits to the doctor. Glycemic stability means fewer dose adjustments and regimens-easier to control treatment.
All of this adds up to a better quality of life and fewer complications in the future.
Clinical studies and meta-analyses confirm:
Dorzagliatin provides a fast start, sustained reduction of HBA₁C, restoration of beta-cell function and a high safety profile – both in monotherapy and in combination with metformin.
For the doctor, it is a modern tool for improving control in patients with insufficient compensation.
This is a real chance for the patient to feel better, control their blood sugar, and reduce their risks without complex or aggressive therapy.
Sources:
https://www.frontiersin.org
https://onlinelibrary.wiley.com
https://pubmed.ncbi.nlm.nih.gov
https://www.nature.com